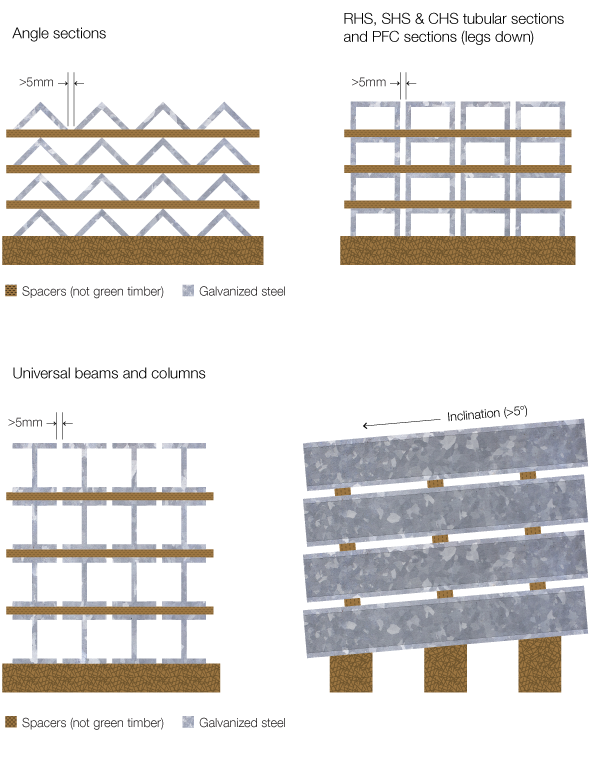
Protective life of galvanized coating
Corrosivity in the Australian and New Zealand atmosphere
Three key Standards, AS/NZS 2312.2, AS 4312 and SNZ TS 3404, provide considerable information on the corrosion rate of steel and zinc under various conditions of atmospheric service. In addition, over the last few decades the CSIRO, HERA and others have carried out extensive mapping to establish the corrosivity of the Australian and New Zealand climate. AS/NZS 2312.2 is the best source for the durability of galvanized coatings in Australian and New Zealand conditions, AS 4312 is the best source of Australian data for corrosivity of steel in the local environment, while SNZ TS 3404 provides corrosion maps for all New Zealand and is a core reference for durability design in New Zealand.
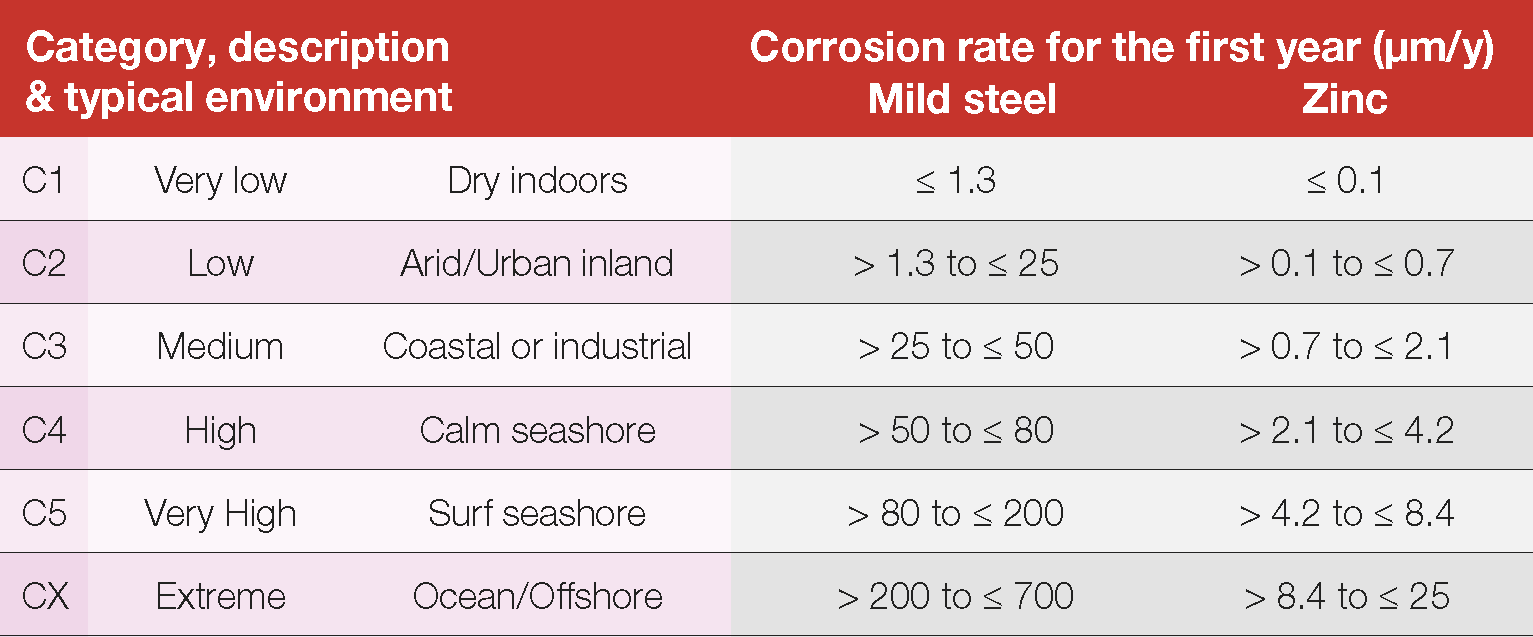
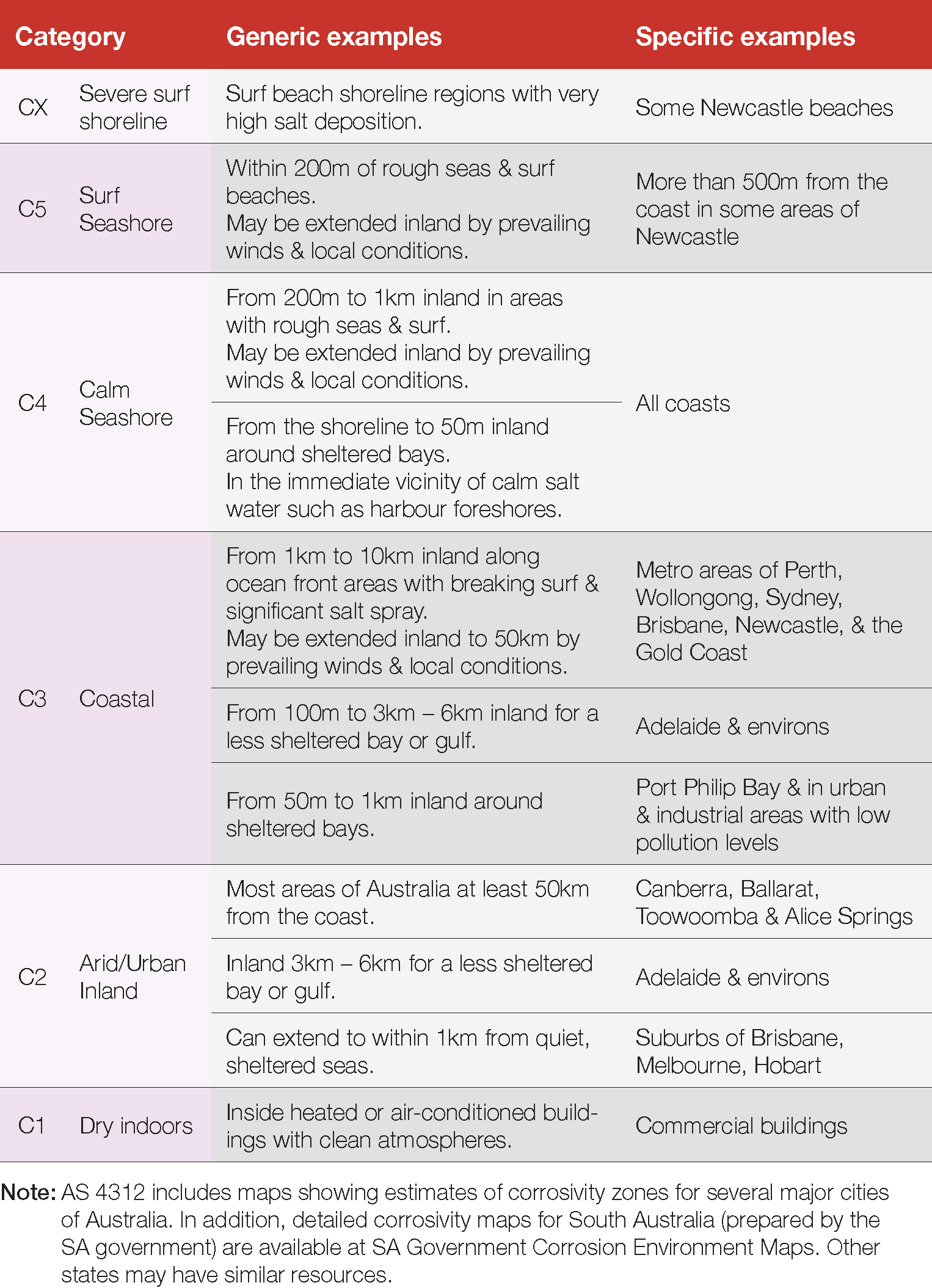
The corrosion rate estimates for steel and zinc in the various local Standards are consistent and are interchangeable with the International Standard ISO 9223 for service life estimate purposes. For ease of design the environments are organised into Corrosivity Categories (C1 – CX) and corrosion rate ranges for zinc and steel are provided in the Standards (Table 1). Table 2 and Figure 18 provide more information on typical environments.
Table 1 Corrosion rates of steel & zinc from ISO 9223 (identical to AS 4312 and AS/NZS 2312.2)
The measured corrosion rate during the first year of exposure for steel and zinc is usually higher than the measured long-term rate. The long-term corrosion rate for both steel and zinc slows down over time and in the first 20 years corrosion rates are not linear. At some point in time after 20 years the corrosion product layer stabilises, and at this point, the corrosion rate becomes linear with time, because the rate of metal loss becomes equal to the rate of loss from the corrosion product layer. In addition, it is known that the rate of corrosion in the first 12 months is strongly affected by the environment experienced by the metal in the first few weeks of exposure, but long-term rates are usually unaffected by the first exposure rate. Notwithstanding the issues outlined above, the durability of an item is usually calculated on the estimated first year corrosion rate and this will normally provide a conservative prediction of the durability of an item if long term corrosion protection is desired.
The estimation of the protective life of a galvanized coating in a location must consider factors such as climatic conditions, the presence of contaminants in the atmosphere introduced by urban or industrial activity, and chlorides in the air which are generally related to proximity to the sea. For more information on how these factors affect the durability of the hot dip galvanized coating, see the GAA Guide to the Service Life of Galvanizing.
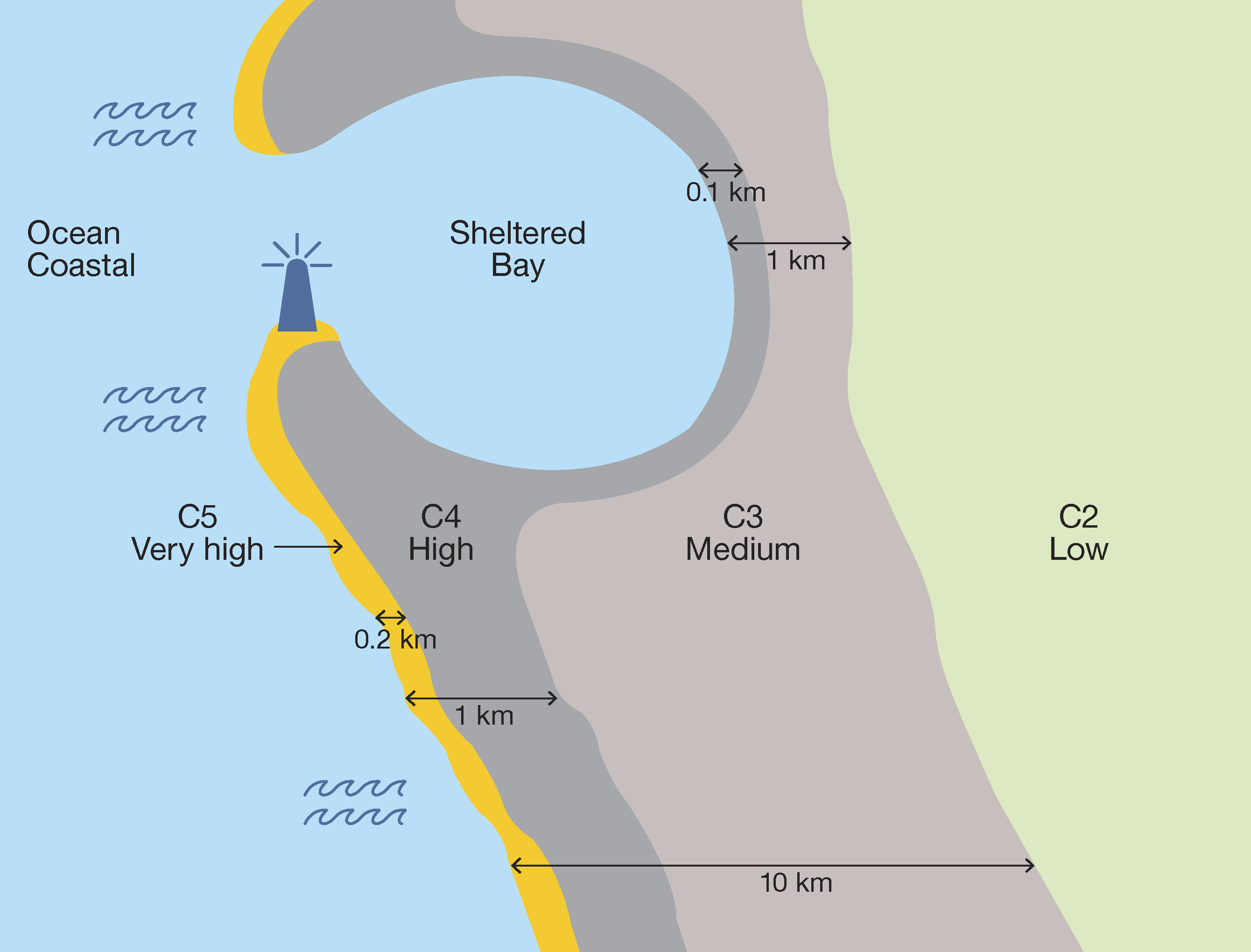
Some environments which appear similar can produce considerable differences in corrosivity due to relativity minor variations in conditions, such as prevailing winds, proximity to corrosive influences, and general atmospheric conditions.
In warm dry atmospheres zinc is very stable. The patina formed during initial exposure remains intact preventing further reaction between the galvanized coating and the air, and protection continues indefinitely.
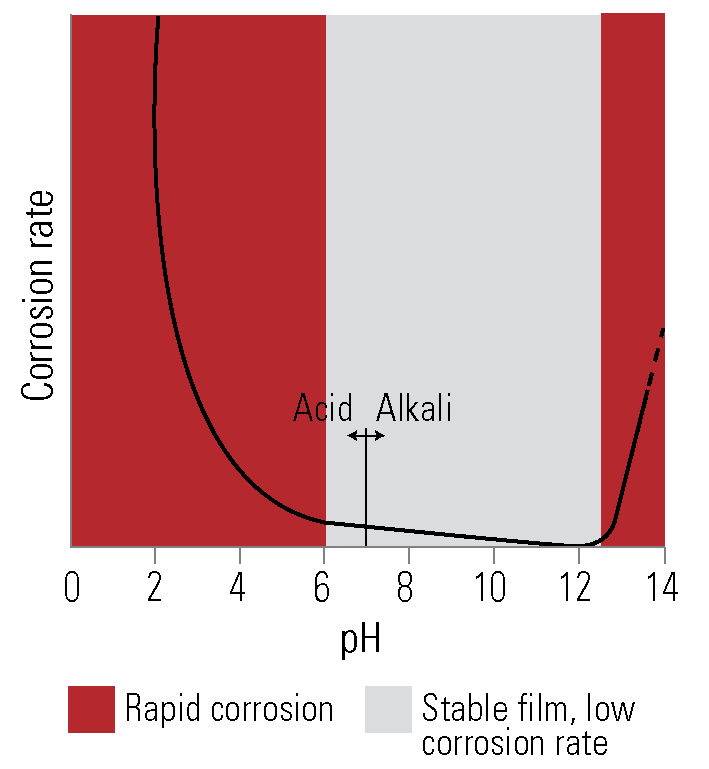
In the presence of atmospheric moisture, the zinc oxide film is quickly converted to zinc hydroxide, and carbon dioxide present in the air reacts to form basic zinc carbonates. These stable inert compounds resist further action and ensure long life for the protective galvanized coating.
In rural areas the life of galvanized coatings is often over 100 years but may be reduced by micro-environments created by the effects of aerial spraying of fertilizers or insecticides. In dry form, most fertilizers and insecticides are harmless to zinc coatings but when wetted by rainwater or irrigation spray water, aggressive solutions can be formed which will attack galvanized coatings until washed off by further wetting.
Near the seacoast the rate of corrosion is increased by the presence of soluble chlorides in the atmosphere. Surf coasts and crashing surf are likely to significantly reduce the life of a galvanized coating, while calm coasts are generally relatively benign once the galvanized steel is more than 50m from the high tide. Micro-environments play a critical part in determining the life of a galvanized coating in these areas, with building density and location, trees and other foliage and topography playing a significant role in increasing or decreasing the corrosiveness. The performance of galvanized coatings relative to other protective systems is outstanding, particularly when used as part of a duplex system consisting of paint over galvanizing.
In the tropical regions and away from surf coasts, the corrosivity, while initially CX to C5 at the high tide line, drops dramatically to C2 within 1 kilometre and hot dip galvanized steel coatings usually significantly outperform organic coatings in these locations.
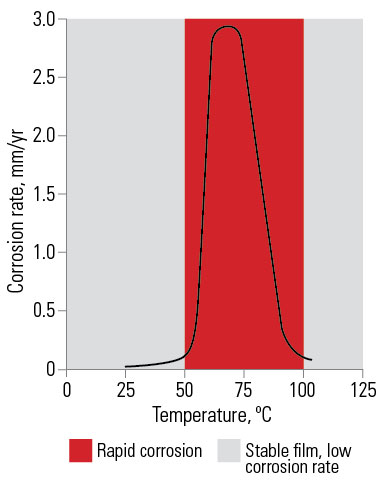
In industrial areas and the volcanic region of New Zealand the presence of atmospheric impurities such as sulphurous gases and chemicals results in the formation of readily soluble zinc salts. These are removed by moisture, which then exposes more zinc to attack. In light industrial areas, galvanized coatings give adequate protection, but in the extremely corrosive conditions of heavy industrial areas and nearby to the volcanic regions it is desirable to reinforce galvanized coatings with a paint system resistant to the chemical attack.
Table 2 Corrosivity in Australia as described in AS 4312
How service life relates to coating thickness
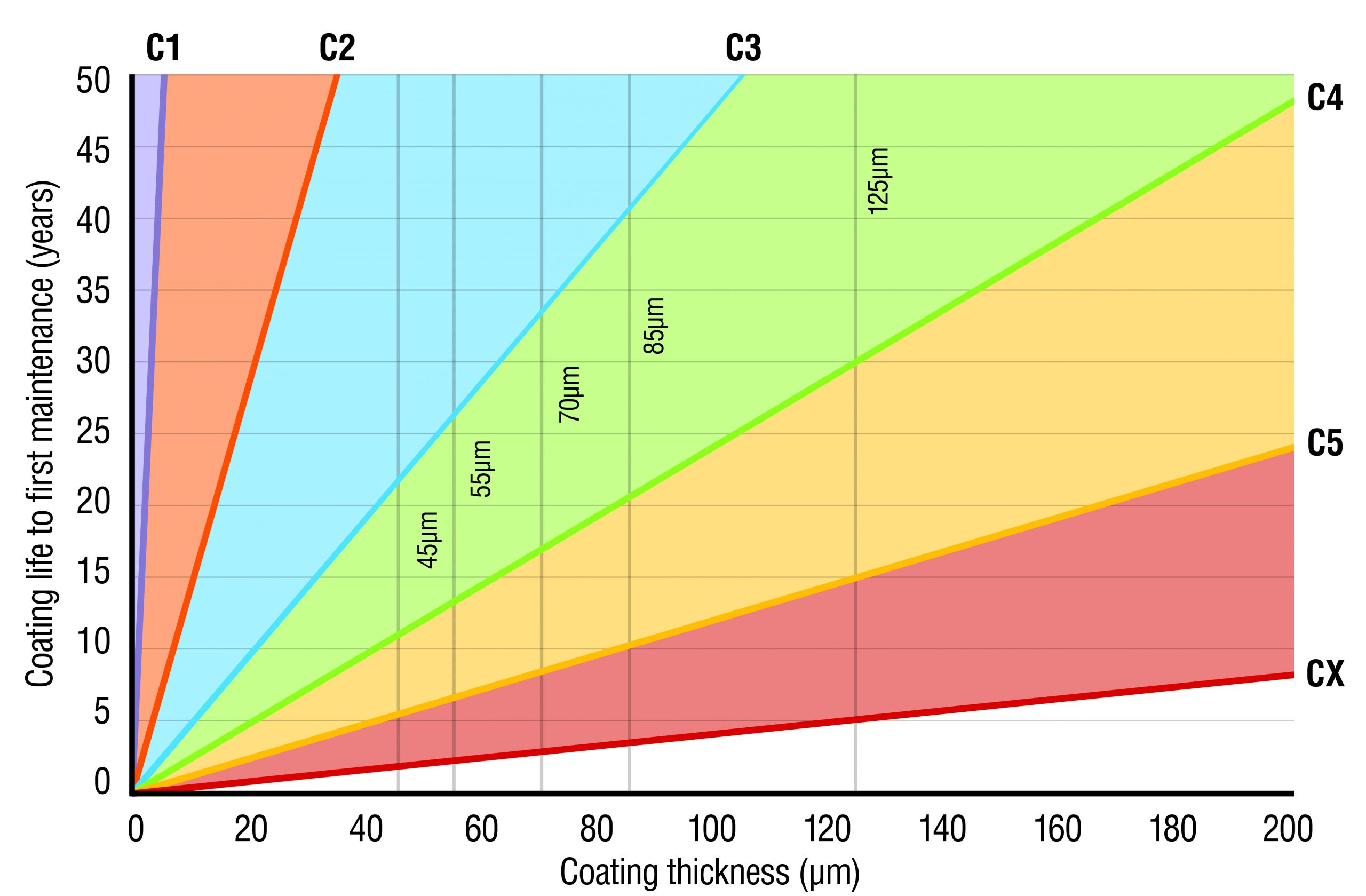
The service life of any post-fabrication galvanized item is directly proportional to the thickness of the coating. This in turn is a function of the thickness of the metal; the thicker the steel, the thicker the layer of zinc (Table 3 of Process section).
Indeed, one of the great advantages of hot dip galvanizing is the predictability of the thickness of the zinc for any given steel thickness. This is particularly important for sharp edges and complex shapes, where conventional paints do not always cover well.
Continuous galvanizing and electro-galvanizing processes do not follow this relationship, because the processes deliberately restrict the galvanizing thickness to allow the steel to retain ductility for further manufacturing.
Durability of commonly available galvanized coatings
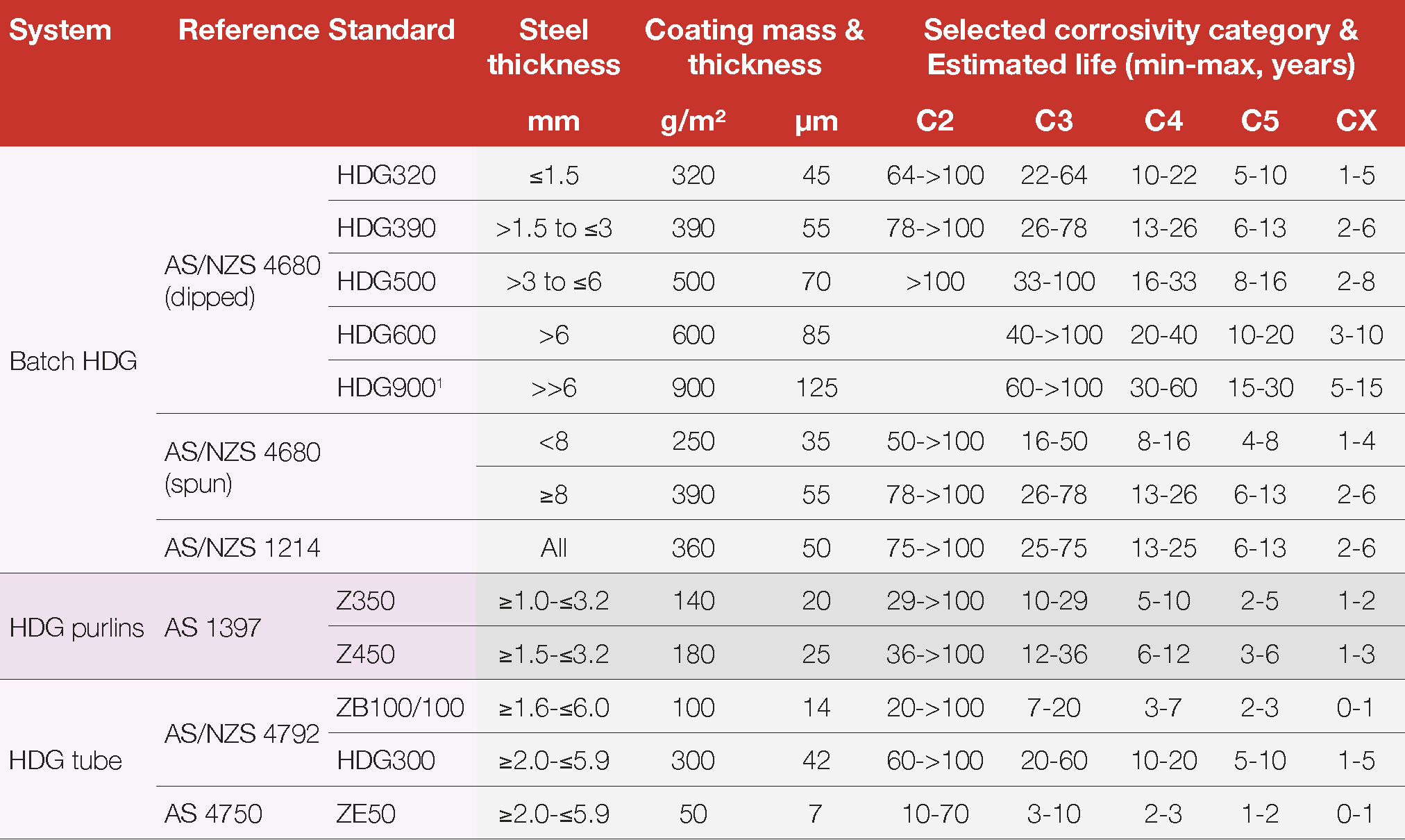
There are many different hot dip galvanized coatings available in Australia and New Zealand. Some commonly available products and their relative performance in various environments are shown in Table 3. It is important to recognise that the durability of all galvanized and other zinc coatings is generally directly proportional to the coating thickness, which is defined in each manufacturing Standard.
After-fabrication hot dip galvanizing manufactured to AS/NZS 4680 produces the maximum thickness possible relative to steel thickness, which means it will always provide a longer life to first maintenance than any other hot dip galvanized coating. This contrasts with continuously or specialised coated products, which are produced by different processes. For continuous coated products, the zinc thickness is accurately controlled, regardless of steel thickness, so that it remains smooth and ductile to allow for later cold forming and for varying manufacturing and end usage and therefore the corrosion protection is necessarily less because of the lower zinc thickness.
The GAA has produced an on-line estimator for the protective life of galvanized coatings based on AS/NZS 2312.2 and common hot dip galvanizing standards. It can be freely accessed here: https://gaa.com.au/durability-of-galvanizing-estimator/.
Table 3 Estimated life to first maintenance for commonly available hot dip galvanized products
Notes:
- Hot dip galvanized coatings thicker than 85µm are not specified in AS/NZS 4680 but the general provisions of that Standard apply and, together with specific thickness figures, may form a specification capable of third-party verification. It is essential to know the composition of the steel to be used and the galvanizer should be consulted before specifying, as these thicker coatings may not be available for all types of steel. Where the steel is suitable, thick coatings may be specified.
- AS 1397 C350 and C450 are typically supplied as purlins (See for example, Lysaght, Stramit, Fielders, etc.). The base steel thicknesses quoted here is the usual range used for these products. Other coating thicknesses may be available, but the coating thickness supplied is independent of the base steel thickness.
- AS/NZS 4792 ZB100/100 is the usual specification of the coating supplied by Australian Tube Mills for their DuraGalPLUS® range of tubular products. Thicker coatings (ZB 135/135) may be available for a limited range of specific tubular building products.
- AS/NZS 4792 HDG300 is the typical Australian Tube Mills and Orrcon specification for specialised HDG coatings applied to circular hollow sections.
- AS 4750 ZE50 is the usual specification of the coating supplied by Orrcon for their ALLGAL® range of tubular products.
The estimated zinc corrosion rates can also be displayed graphically, allowing for simple calculations for Life to First Maintenance based on measured coating thicknesses.
Statement of Performance
Members of the Galvanizers Association of Australia (GAA) supply the service of hot dip galvanizing on fabricated articles to AS/NZS 4680, unless otherwise agreed in writing.
The durability of the finished hot dip galvanized coating is a function of the environment to which the article is exposed. For atmospheric environments common to Australia, this durability can be estimated from AS/NZS 2312.2 (see https://gaa.com.au/durability-of-galvanizing-estimator/).
The galvanizer has no control over the final location and installation of the hot dip galvanized article, nor its intended use or maintenance of the finished hot dip galvanized coating which must necessarily be carried out by the end user. Therefore, the galvanizer has no control over the durability of the hot dip galvanized coating.
A declaration of conformance to AS/NZS 4680 is available from all members of the GAA on request.
If requested and agreed in writing prior to galvanizing of the fabricated article, a test certificate showing the measured galvanized coating thickness from a sample inspection carried out to the requirements of AS/NZS 4680 is available from all members of the GAA.